mGluR6 trafficking and internalization
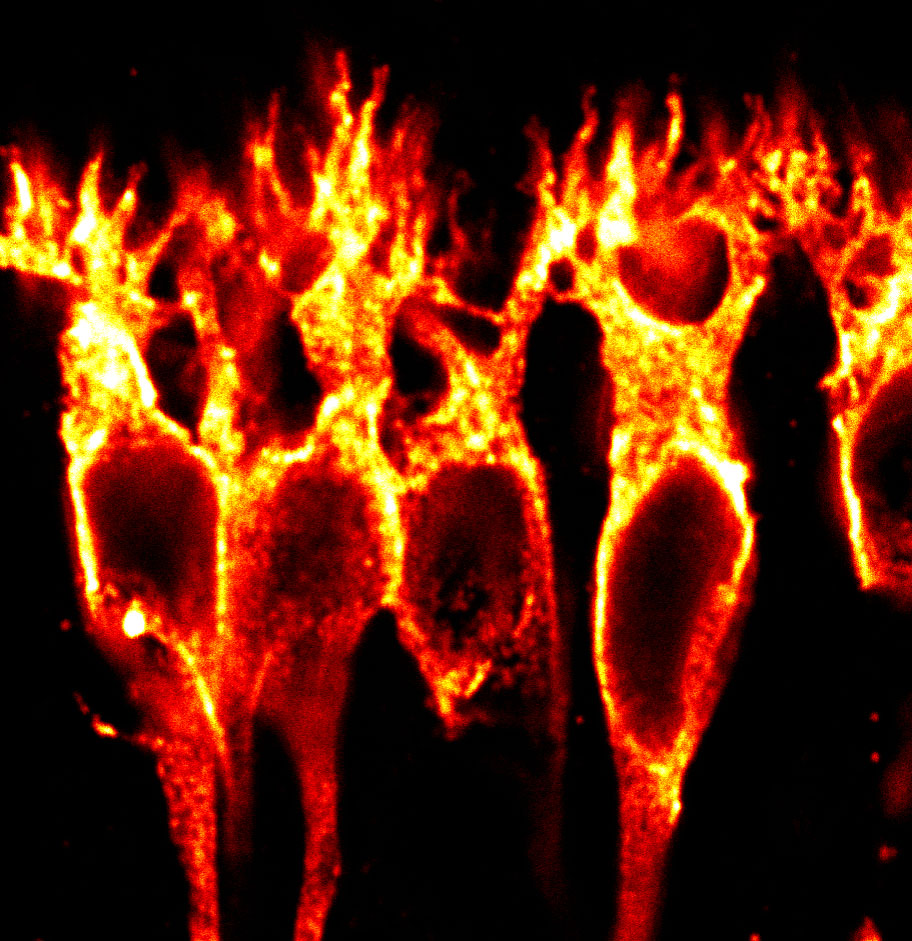
At rod photoreceptor synapses in the retina, glutamate released from rods is detected by mGluR6 at bipolar cell dendritic tips. We previously reported that mGluR6 is present not only at bipolar cell dendritic tips, but also in an intracellular pool in bipolar cell somas (1). The mechanisms that regulate trafficking and plasma membrane insertion of mGluR6 are unknown.
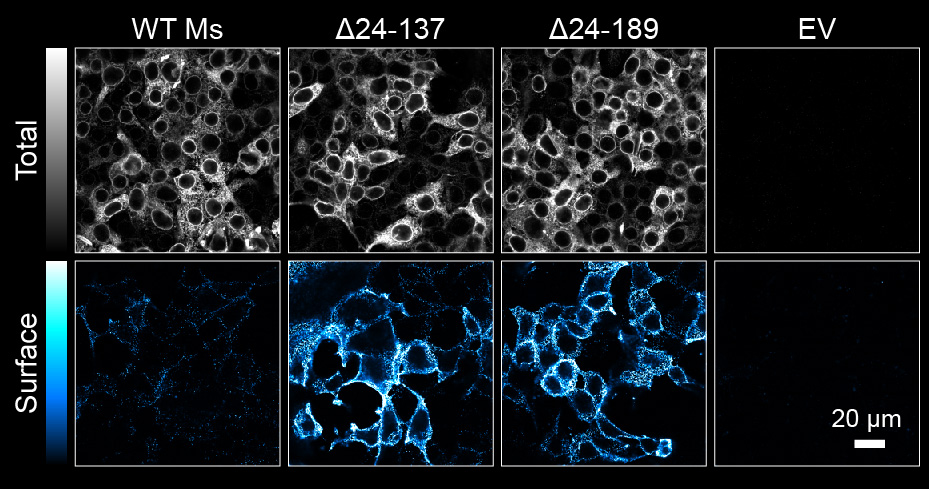
We reported that deletions of large portions of the mGluR6 extracellular ligand-binding domain lead to 10-20 fold increases in surface expression in heterologous cells and glycosylation defects (Fig 1, 2) (1,2). Ongoing projects are examining the role of the mGluR6 extracellular domain in regulating secretory trafficking.
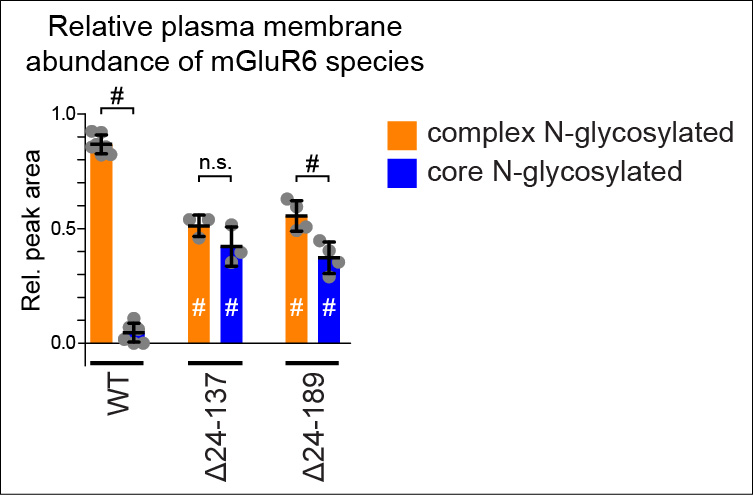
Fig 2. Plasma membrane insertion of immature core glycosylated deletion mutants in HEK cells. Adapted from (2).
Using an N-terminal SNAP tag, we can visualize mGluR6 internalization in heterologous cells (Fig 3) (2). Ongoing projects are examining the mechanisms of mGluR6 internalization.
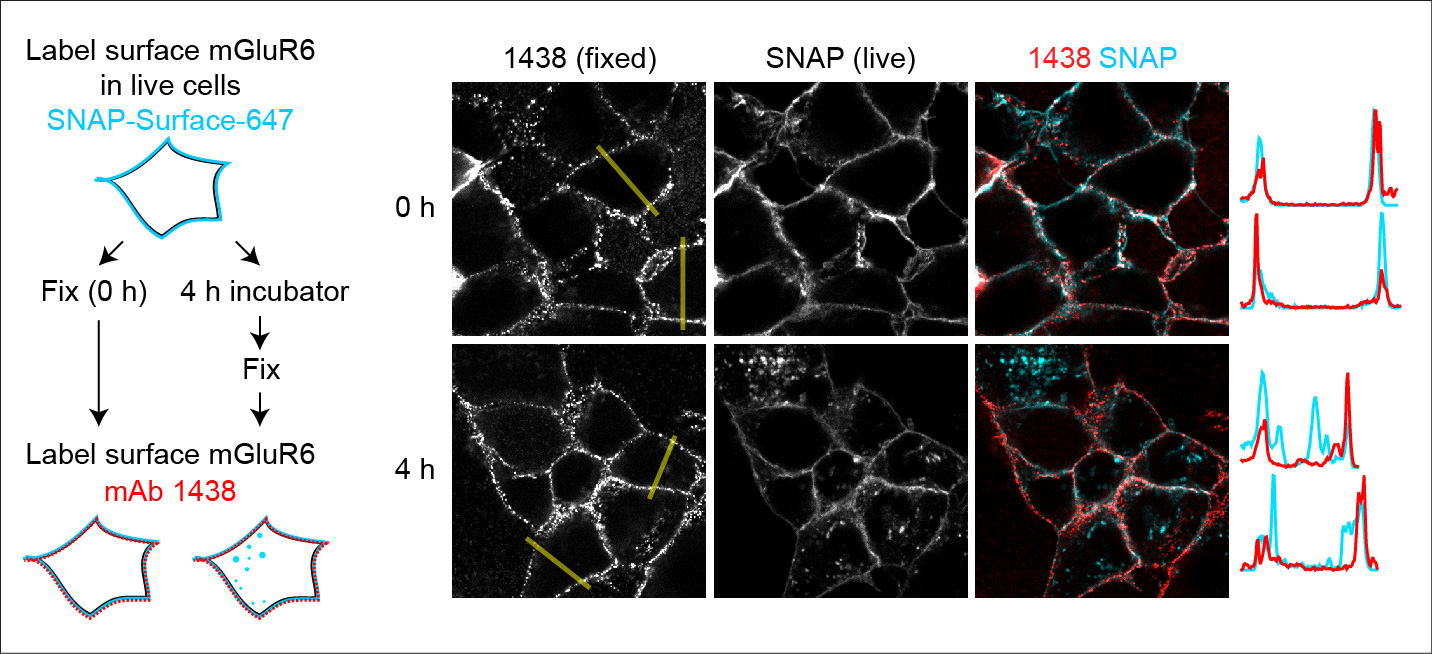
Fig 3. Live cells were labeled with fluorescent SNAP ligand. After four hours, internalization of labeled proteins can be detected. Adapted from (2).
References
1. Agosto MA, Adeosun AA, Kumar N, Wensel TG. 2021. The mGluR6 ligand-binding domain, but not the C-terminal domain, is required for synaptic localization in retinal ON-bipolar cells. J Biol Chem 297, 101418.
2. Rideout AP, Abuelatta OW, Agosto MA. 2026. The extracellular domain of mGluR6 regulates targeting to the conventional secretion pathway. Mol Cell Neurosci 136, 104068