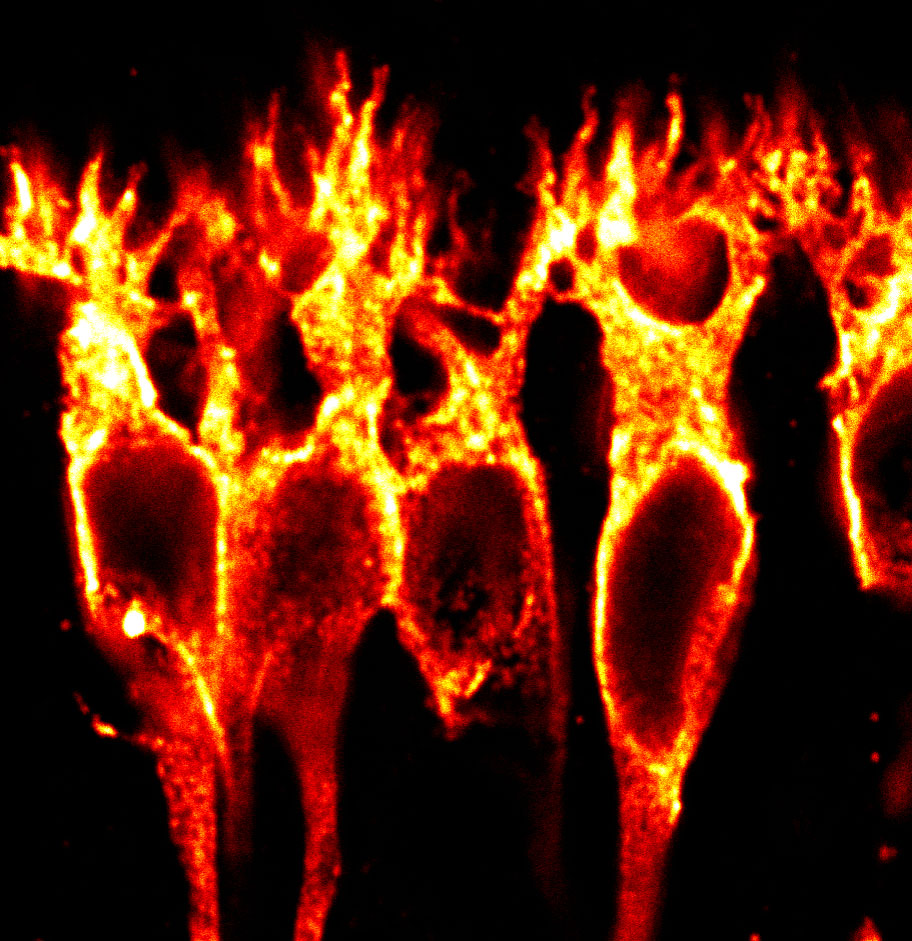
mGluR6 glycosylation and ELFN complexes
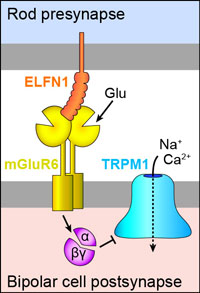
Fig 1. Diagram of a synapse between a rod photoreceptor and rod bipolar cell.
Metabotropic glutamate receptors (mGluRs) are G-protein coupled receptors that are activated by glutamate. A subset of mGluRs (mGluR4, mGluR6, mGluR7, and mGluR8) can interact trans-synaptically with ELFN proteins, and these interactions affect synapse formation and function (1–5). At rod photoreceptor synapses in the retina, glutamate released from rods is detected by mGluR6 at bipolar cell dendritic tips (Fig 1).
Localization of mGluR6 at rod bipolar cell dendritic tips depends on a trans-synaptic interaction with presynaptic ELFN1 (2). We showed that this interaction requires N-linked glycosylation of mGluR6, and specifically a complex form of glycosylation acquired in the Golgi during secretory trafficking (Fig 2) (6). Mutation of glycosylation sites in mGluR6 abolished both ELFN1 binding in vitro and synaptic localization in retinal bipolar cells (6).
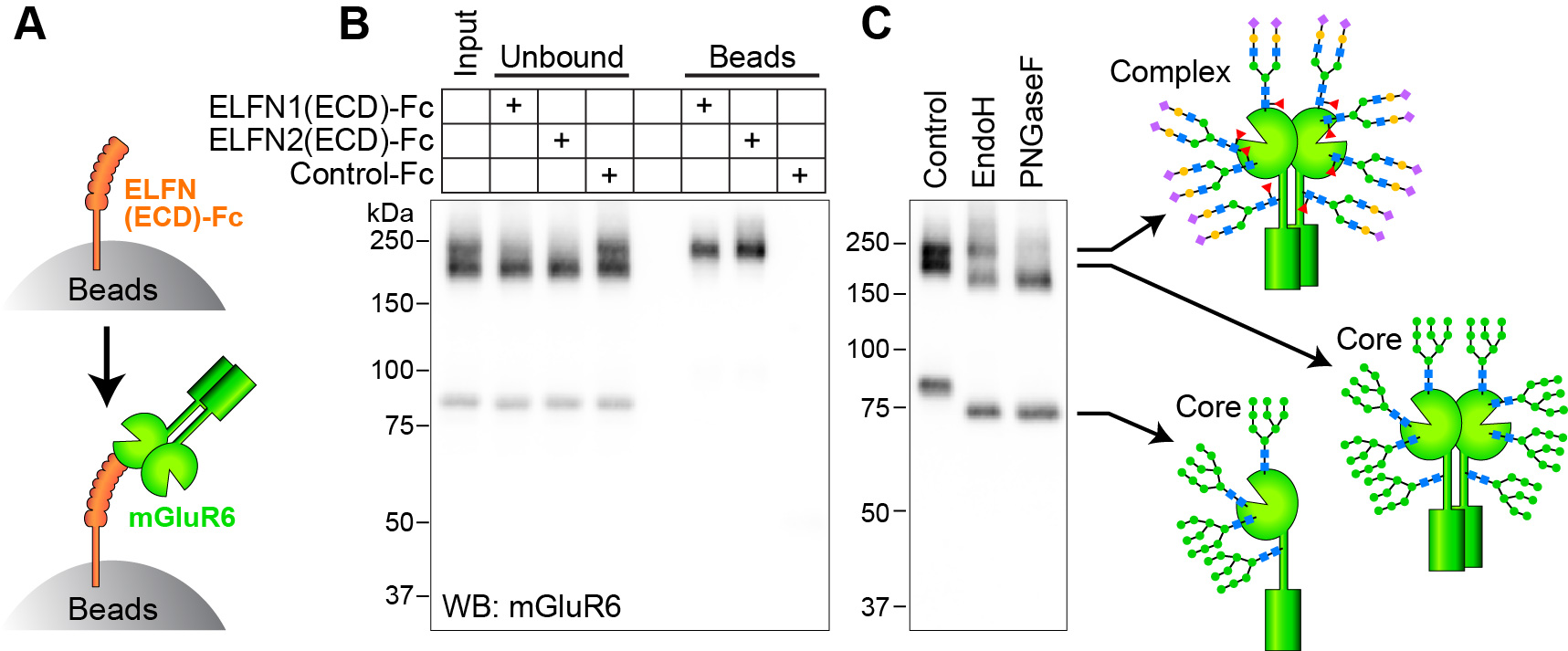
Fig 2. (A) Diagram of the binding assay using ELFN extracellular domain (ECD) fused to Fc. (B) ELFN1 and ELFN2 selectively pull down the upper-most mGluR6 band. (C) The upper band was identified as a complex-glycosylated species using glycosidase enzymes Endo H, which removes only core glycans, and PNGase F, which removes all N-linked glycans. Adapted from (6).
Congenital stationary night blindness (CSNB) is a disease resulting from dysfunction of synaptic transmission between photoreceptors and bipolar cells. Alleles of numerous synaptic proteins, including mGluR6, are associated with CSNB (7). We found that several mGluR6 CSNB variants with point mutations in the extracellular part of the protein had glycosylation defects (8). These mGluR6 variants were unable to bind ELFN1 and were mislocalized in bipolar cells (Fig 3), explaining the loss of function in CSNB (8).
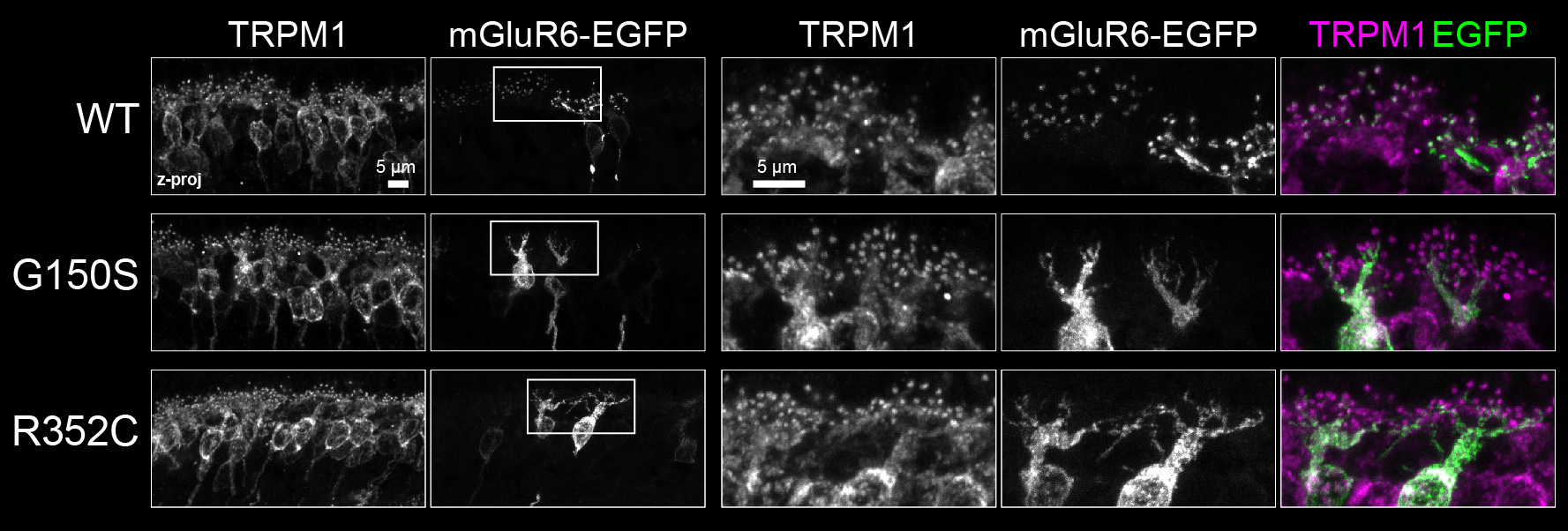
Fig 3. Localization of WT and mutant human mGluR6-EGFP in mouse retinal bipolar cells. Box shows zoomed in region at right. Two examples of mislocalized CSNB variants are shown. Adapted from (8).
We are continuing this project with the aim of dissecting the role of glycosylation in mGluR trafficking and function, specific requirements for ELFN binding, and dysfunction of CSNB variants.
References
1. Tomioka NH, Yasuda H, Miyamoto H, et al. 2014. Elfn1 recruits presynaptic mGluR7 in trans and its loss results in seizures. Nat Commun 5, 1–16.
2. Cao Y, Sarria I, Fehlhaber KE, et al. 2015. Mechanism for selective synaptic wiring of rod photoreceptors into the retinal circuitry and its role in vision. Neuron 87, 1248–1260.
3. Dunn HA, Patil DN, Cao Y, et al. 2018. Synaptic adhesion protein ELFN1 is a selective allosteric modulator of group III metabotropic glutamate receptors in trans. Proc Natl Acad Sci USA 115, 5022–5027.
4. Stachniak TJ, Sylwestrak EL, Scheiffele P, et al. 2019. Elfn1-induced constitutive activation of mGluR7 determines frequency-dependent recruitment of somatostatin interneurons. J Neurosci 39, 4461–4474.
5. Dunn HA, Zucca S, Dao M, et al. 2019. ELFN2 is a postsynaptic cell adhesion molecule with essential roles in controlling group III mGluRs in the brain and neuropsychiatric behavior. Mol Psychiatry 24, 1902–1919.
6. Miller ML, Pindwarawala M, Agosto MA. 2024. Complex N-glycosylation of mGluR6 is required for trans-synaptic interaction with ELFN adhesion proteins. J Biol Chem 300, 107119.
7. Zeitz C, Robson AG, Audo I. 2015. Congenital stationary night blindness: an analysis and update of genotype-phenotype correlations and pathogenic mechanisms. Prog Retin Eye Res 45, 58–110.
8. Pindwarawala M, Abid FA, Lee J, et al. 2024. Defective glycosylation and ELFN1 binding of mGluR6 congenital stationary night blindness mutants. Life Sci Alliance 8, e202403118.